Start with OPSUMIT® (macitentan) to reduce the risks of disease progression and hospitalization for PAH1
For the treatment of pulmonary arterial hypertension (PAH, WHO Group I, Functional Class II-III)
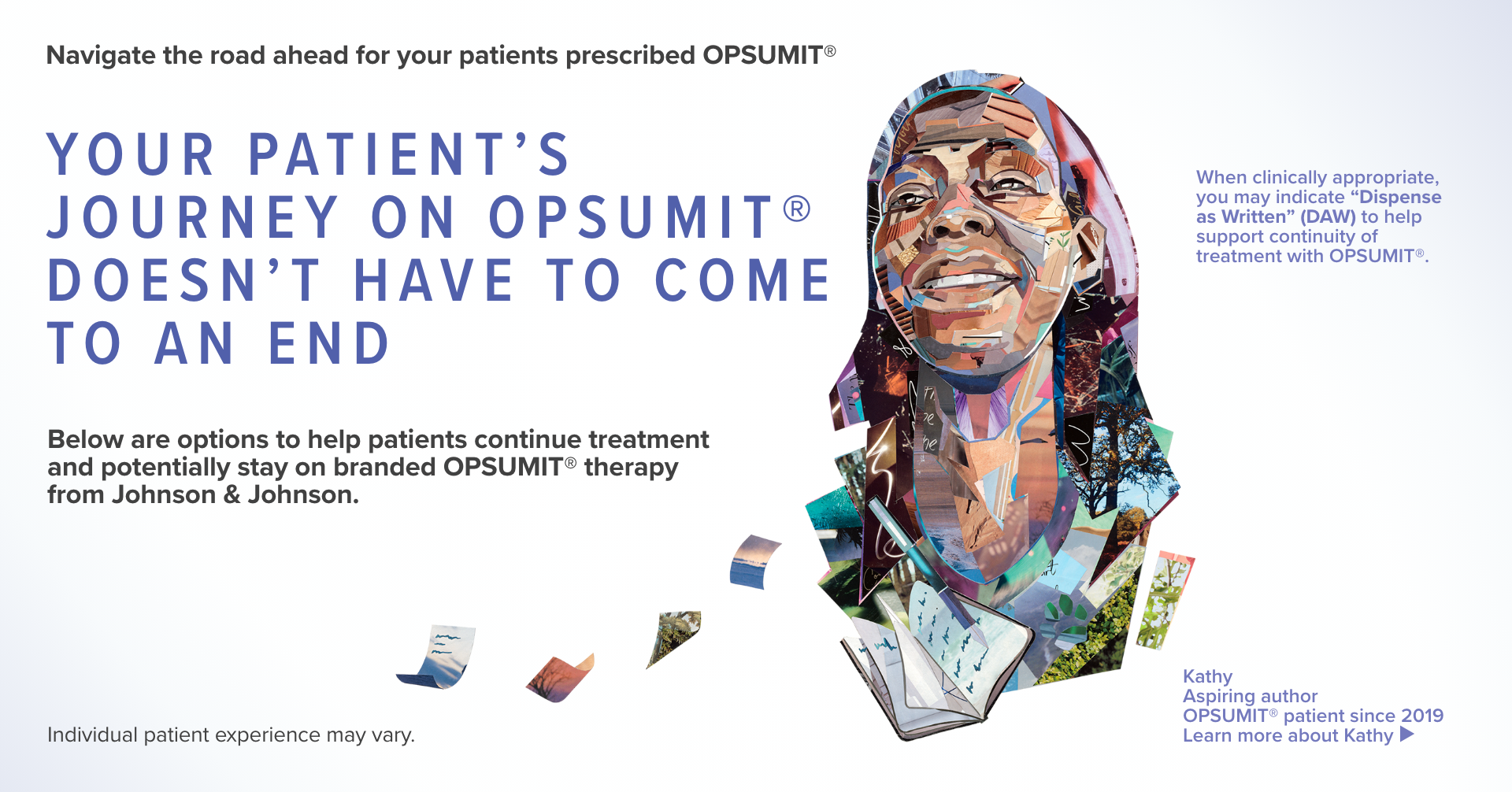
When clinically appropriate, you may indicate “Dispense as Written” (DAW) for patients who maintain coverage for OPSUMIT®.
It may be as easy as checking the DAW box:
- On your prescription pad or e-prescribing platform
- Verbally requesting the prescription be dispensed as written
Patients prescribed OPSUMIT® will continue to have the following J&J withMe resources available.
Therapy Access Managers are also available to help provide access, affordability, and treatment support.
- J&J withMe Oral PAH Savings Program
- J&J withMe Care Coordinators
J&J has a portfolio of PAH therapies. If you are interested in learning more about other options in the portfolio or decide to prescribe another J&J PAH medicine, please speak to your J&J Sales Representative.
*Source: IQVIA NPA TRx (Q1 2023-Q4 2023) and US claims data based on total prescriptions and refills from January 2023 to December 2023 and October 2022 to September 2023, demonstrating that OPSUMIT® is the #1 most-prescribed branded ERA therapy in PAH Nationwide.
HCP=healthcare professional;
PAH=pulmonary arterial hypertension;
PAH-CTD=PAH associated with connective tissue disease;
REPAIR=Right vEntricular remodeling in Pulmonary ArterIal hypeRtension;
SERAPHIN=Study with an Endothelin Receptor Antagonist in Pulmonary Arterial Hypertension to Improve CliNical
Outcome;
WHO=World Health Organization.
References:
1. OPSUMIT® (macitentan) full Prescribing Information. Actelion Pharmaceuticals US, Inc.
2. Data on file (December 2023). Actelion Pharmaceuticals US, Inc. Based on Express Scripts Pharmacy Benefits Manager Claims Data (from January 2023 to December 2023), Optum Clinformatics Extended Data Mart (from October 2022 to September 2023), proprietary claims data (from November 2022 to October 2023), and IQVIA NPA TRx Data (January 2023 to December 2023).







