For U.S. Healthcare Professionals
PAH-related hospitalization
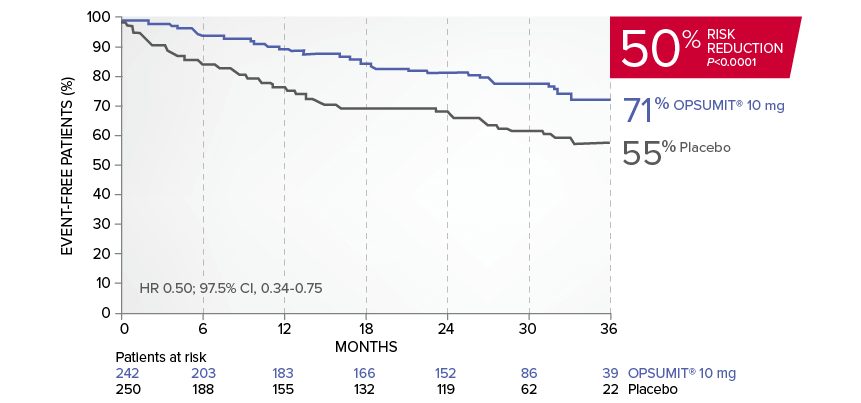
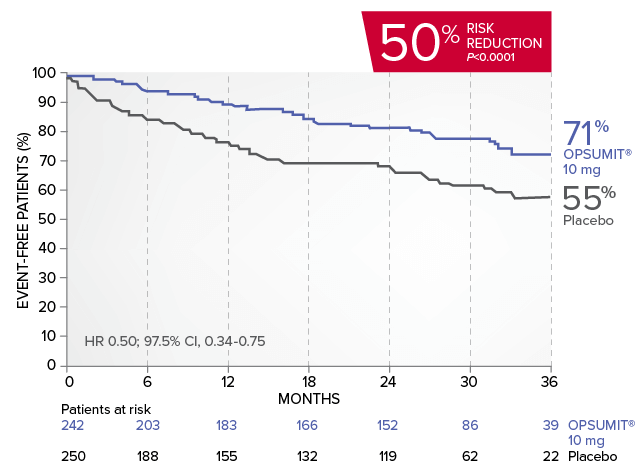
OPSUMIT® (macitentan) reduced the risk of PAH-related hospitalization1,2
A key secondary endpoint in SERAPHIN was death due to PAH or PAH-related hospitalization.
There was a 50% reduction in the risk of PAH-related hospitalization vs placebo in the overall population.*
Kaplan-Meier estimates of risk of first key secondary endpoint event1,2
Summary of death due to PAH and hospitalization due to PAH1,2
| OPSUMIT® 10 mg (n=242), n (%) |
Placebo (n=250), n (%) |
|
|---|---|---|
| Death due to PAH or hospitalization for PAH | 50 (20.7) | 84 (33.6) |
| Component as first event | ||
| Death due to PAH | 5 (2.1) | 5 (2.0) |
| Hospitalization for PAH | 45 (18.6) | 79 (31.6) |
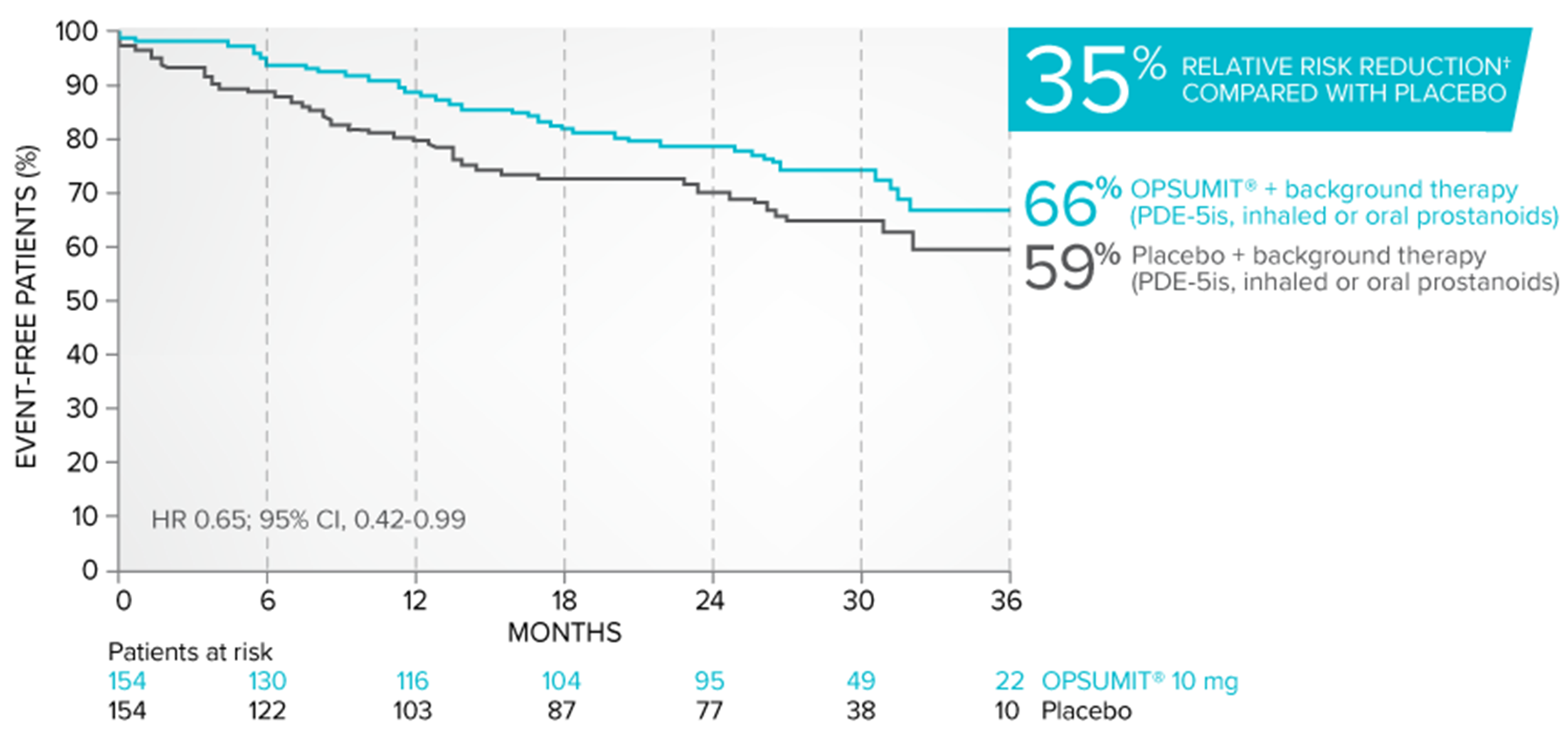
Exploratory subgroup analysis: Combination therapy results
The combination therapy data below represent an exploratory analysis. Results should be interpreted with caution.
Kaplan-Meier estimates of risk of first key secondary endpoint event when OPSUMIT® was added to stable PAH-specific background therapy3
- At baseline, 64% of enrolled patients were treated with a stable dose of PAH-specific background therapy (61% PDE-5is; 6% inhaled or oral prostanoids). OPSUMIT® is approved in combination with PDE-5is or inhaled prostanoids, but not oral prostanoids1
Kaplan-Meier estimates of risk of first key secondary endpoint event3
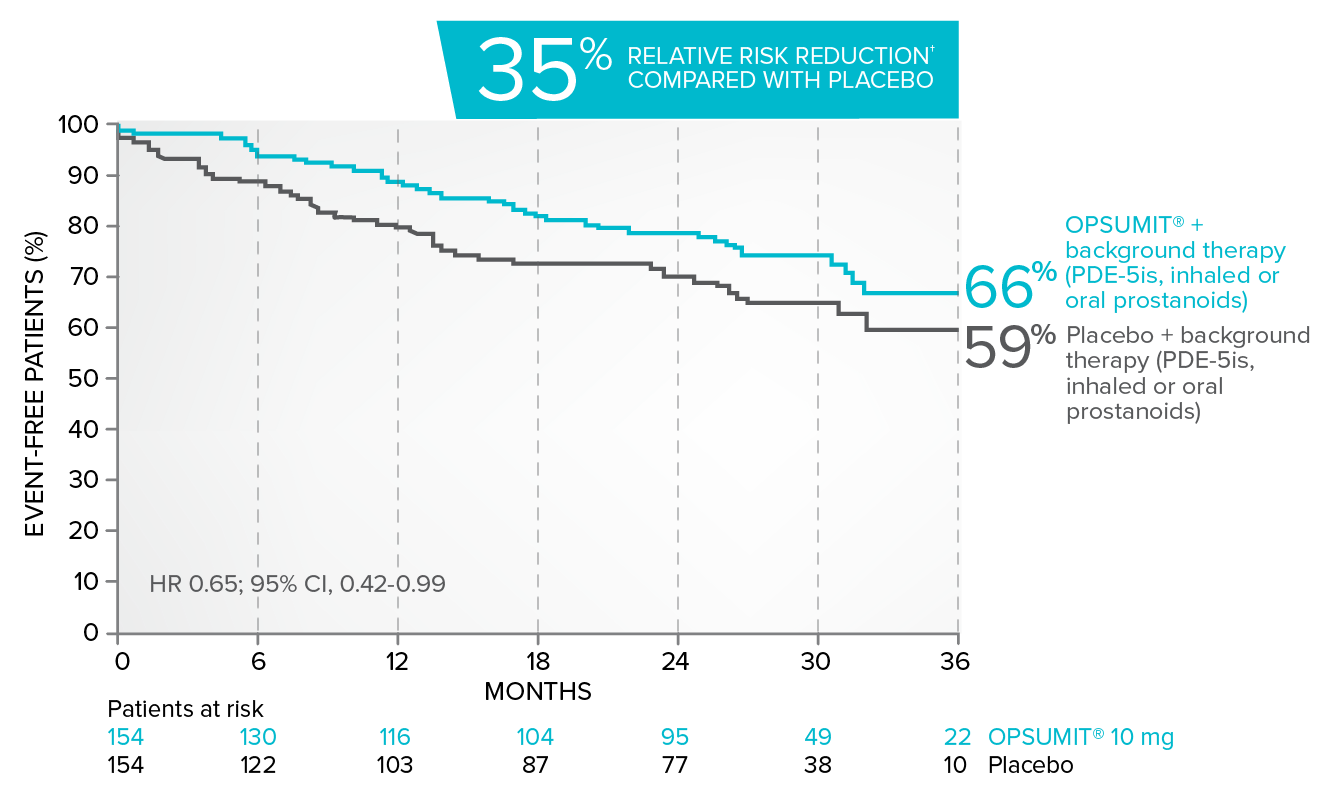
Summary of death due to PAH and hospitalization due to PAH3
| OPSUMIT® 10 mg (n=154), n (%) |
Placebo (n=154), n (%) |
|
|---|---|---|
| Death due to PAH or hospitalization for PAH | 37 (24.0) | 49 (31.8) |
| Component as first event | ||
| Death due to PAH | 2 (1.3) | 2 (1.3) |
| Hospitalization for PAH | 35 (22.7) | 47 (30.5) |
Common adverse reactions in the combination therapy exploratory subgroup4‡
| OPSUMIT® 10 mg n=154 |
Placebo n=153 |
|
|---|---|---|
| Anemia | 16.2% | 4.6% |
| Nasopharyngitis | 11.0% | 10.5% |
| Bronchitis | 11.0% | 5.9% |
| Headache | 13.6% | 10.5% |
| Diarrhea | 13.0% | 9.8% |
‡More frequent than placebo by ≥3%.
Combination therapy exploratory subgroup safety2-4
- The safety profile of OPSUMIT® as part of a combination therapy regimen was consistent with that of OPSUMIT® in the overall SERAPHIN population
- The incidence of peripheral edema, a known ERA-related adverse event, was similar in patients receiving background therapy who were treated with OPSUMIT® and placebo (19.5% and 23.5%, respectively)
- Treatment discontinuations due to adverse events in patients receiving background therapy were similar in those receiving OPSUMIT® and those receiving placebo (9.1% and 11.8%, respectively)
*All randomized patients.
†Not adjusted for multiplicity.
Looking for patient enrollment resources?
Enroll PatientsAccess the
video library
Watch Videos
Review the
REPAIR study
DISCOVER THE DATA
CI=confidence interval;
ERA=endothelin receptor antagonist;
HR=hazard ratio;
PAH=pulmonary arterial hypertension;
PDE-5i=phosphodiesterase type 5 inhibitor;
SERAPHIN=Study with an Endothelin Receptor Antagonist in Pulmonary Arterial Hypertension to Improve CliNical Outcome.
References:
1. OPSUMIT® [prescribing information]. Actelion Pharmaceuticals US, Inc.
2. Pulido T, Adzerikho I, Channick RN, et al; SERAPHIN Investigators. Macitentan and morbidity and mortality in pulmonary arterial hypertension. N Engl J Med. 2013;369(9):809-818 and suppl 1-21. doi:10.1056/NEJMoa1213917
3. Data on file. Actelion Pharmaceuticals US, Inc.
4. Jansa P, Pulido T. Macitentan in pulmonary arterial hypertension: a focus on combination therapy in the SERAPHIN trial. Am J Cardiovasc Drugs. 2018;18:1-11.