For U.S. Healthcare Professionals
OPSUMIT® (macitentan) was studied in SERAPHIN, the largest long-term, outcomes-based pivotal trial of an ERA in PAH1
Long-term outcomes data in both monotherapy AND combination therapy1,2
Trial design1,2:
The effect of OPSUMIT® on disease progression in patients with PAH (WHO Group I) was studied in SERAPHIN, a large (N=742), event-driven, multicenter, long-term (average treatment duration 2 years), randomized, double-blind, placebo-controlled phase 3 trial. At study baseline, 36% of patients were not using PAH-specific background therapy and 64% were using stable background therapy for at least 3 months with PDE-5is or inhaled/oral prostanoids.*
Trial demographics1,3:
- Patients had predominantly WHO FC II (52%) and FC III (46%) symptoms
- Etiologies included IPAH/HPAH (57%), PAH-CTD (31%), PAH-CHD with repaired shunts (8%), PAH associated with drugs and toxins (3%), and PAH-HIV (1%)
- Mean patient age was 46 years, and 77% of patients were female
- 25% of patients were recently diagnosed (<6 months) and 75% were previously diagnosed (≥6 months)
Trial demographics1
742
Patients were randomized
OPSUMIT® 10 mg: n=242
Macitentan 3 mg: n=250
Placebo: n=250
Macitentan 3 mg is not
an approved dose.
Monotherapy and
combination therapy1,3
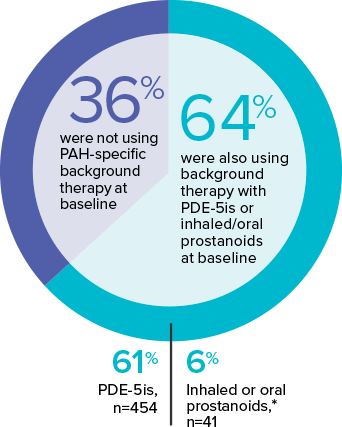
*OPSUMIT® is approved in combination with PDE-5is or inhaled prostanoids, but not oral prostanoids.
Learn about the proven efficacy of OPSUMIT®
See Seraphin ResultsSERAPHIN PAH-related hospitalization results
View the DataView SERAPHIN PAH-CTD subgroup data
See the DataReview the
REPAIR study
DISCOVER THE DATA
ERA=endothelin receptor antagonist;
FC=Functional Class;
HPAH=heritable PAH;
IPAH=idiopathic PAH;
PAH=pulmonary arterial hypertension;
PAH-CHD=PAH associated with congenital heart disease;
PAH-CTD=PAH associated with connective tissue disease;
PAH-HIV=PAH associated with human immunodeficiency virus;
PDE-5i=phosphodiesterase type 5 inhibitor;
SERAPHIN=Study with an Endothelin Receptor Antagonist in Pulmonary Arterial Hypertension to Improve CliNical Outcome;
WHO=World Health Organization.
References:
1. OPSUMIT® [prescribing information]. Actelion Pharmaceuticals US, Inc.
2. Pulido T, Adzerikho I, Channick RN, et al; SERAPHIN Investigators. Macitentan and morbidity and mortality in pulmonary arterial hypertension. N Engl J Med. 2013;369(9):809-818 and suppl 1-21. doi:10.1056/NEJMoa1213917
3. US Dept of Health and Human Services, US Food and Drug Administration, Center for Drug Evaluation and Research. Opsumit® (macitentan) NDA 204410. Accessed October 14, 2022. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2013/204410Orig1s000MedR.pdf